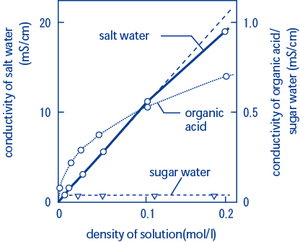
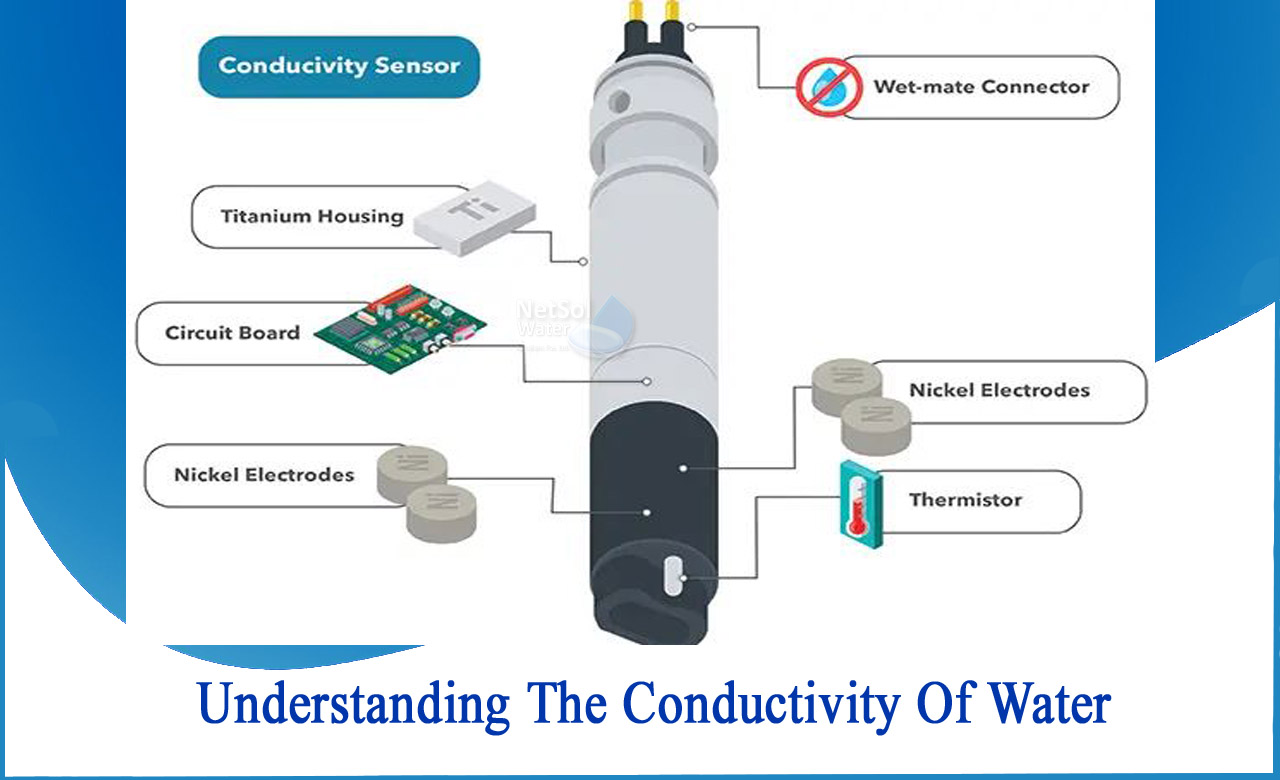
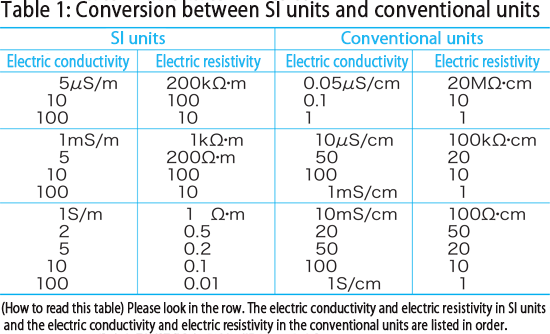
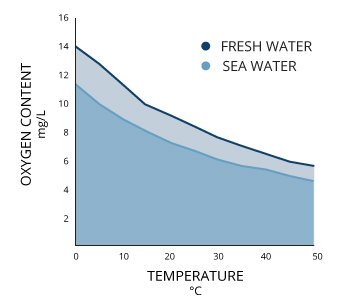
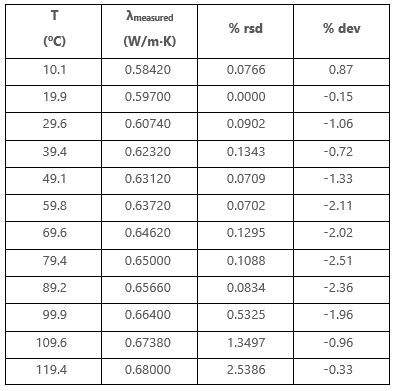
Specific conductance of pure water at `25^(@)C` is `0.58 xx 10^(-7)` mho `cm^(-1)`. Calculate - YouTube

The conductivity of pure water in a conductivity cell with electrodes of cross-sectional area `4cm^( - YouTube

If conductivity of water used to make saturated of AgCl is found to be `3.1xx10^(-5)Omega^(-1)` ` - YouTube
The conductivity of water at 298K is 0.55 × 10^ 7 S cm^ 1. if λ m(H^+)=350 S cm^2 mol^ 1 and λ(OH^ )=200 S cm^2, the degree of dissociation of water will b
At certain temperature the conductivity of pure water is 6.61/100000000ohm cm the ionic conductance of H+ and OH ions at this temperature are 350 and 200ohm 1 cm2 mol 1 respectively the


.webp)

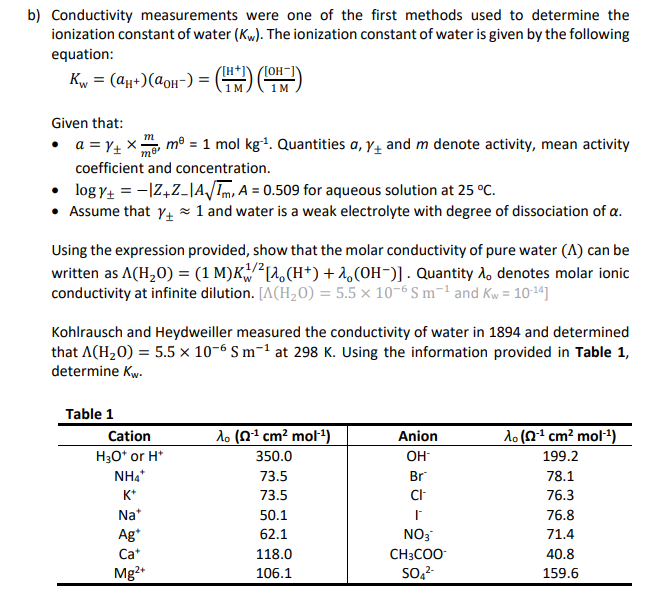

![PDF] Measurements of the electrical conductivity of water | Semantic Scholar PDF] Measurements of the electrical conductivity of water | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/eba8291e5c5479a23830fc6fdfb2652d24031ab5/3-Table1-1.png)